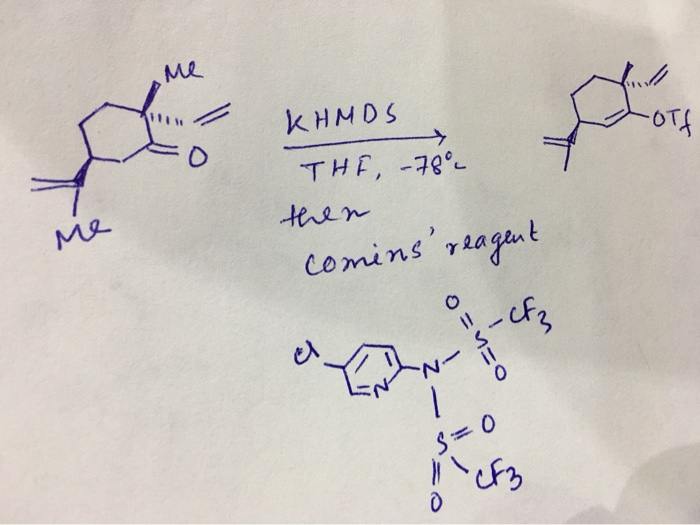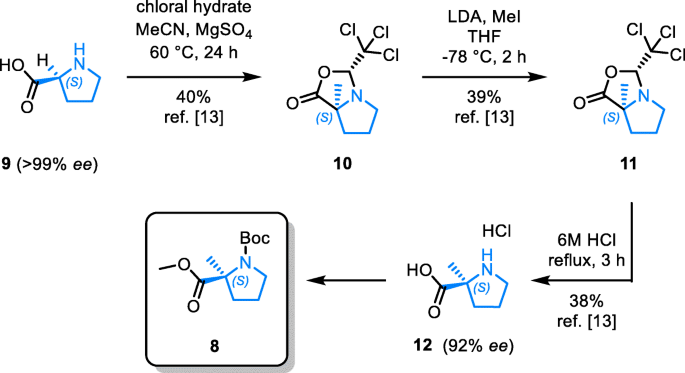
Enantiospecific cyclization of methyl N-(tert-butoxycarbonyl)-N-(3-chloropropyl)-D-alaninate to 2-methylproline derivative via 'memory of chirality' in flow | SpringerLink

Potassium Amide‐Catalyzed Benzylic C−H Bond Addition of Alkylpyridines to Styrenes - Zhai - 2018 - Angewandte Chemie International Edition - Wiley Online Library
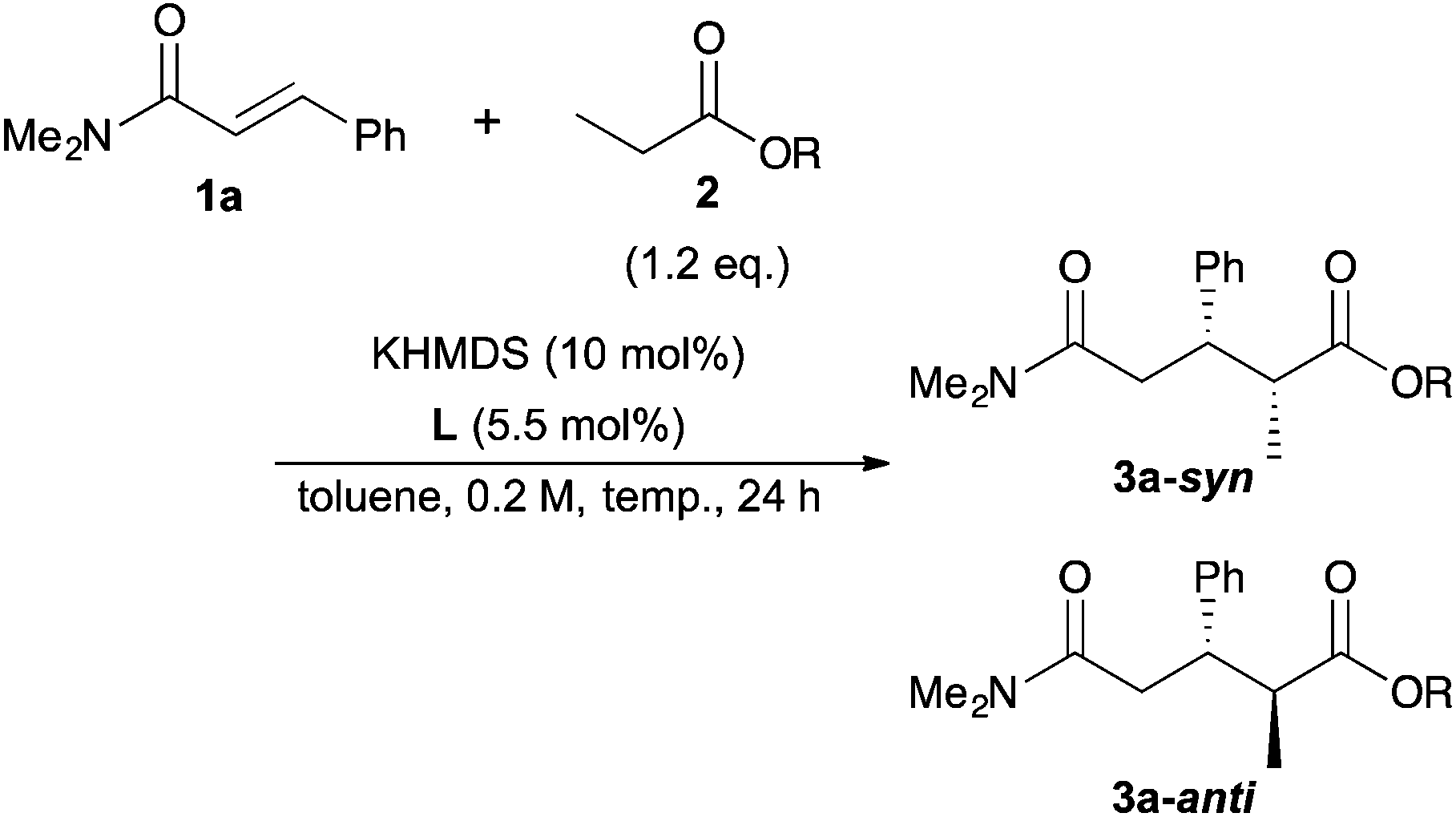
Catalytic asymmetric direct-type 1,4-addition reactions of simple esters - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C6QO00242K
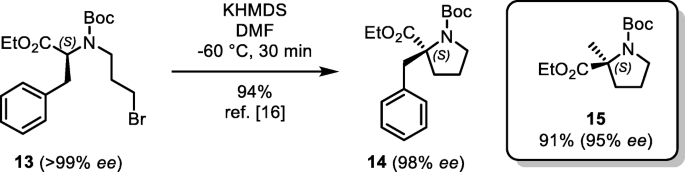
Enantiospecific cyclization of methyl N-(tert-butoxycarbonyl)-N-(3-chloropropyl)-D-alaninate to 2-methylproline derivative via 'memory of chirality' in flow | SpringerLink

Phosphinylation of Non‐activated Aryl Fluorides through Nucleophilic Aromatic Substitution at the Boundary of Concerted and Stepwise Mechanisms - You - 2021 - Angewandte Chemie International Edition - Wiley Online Library
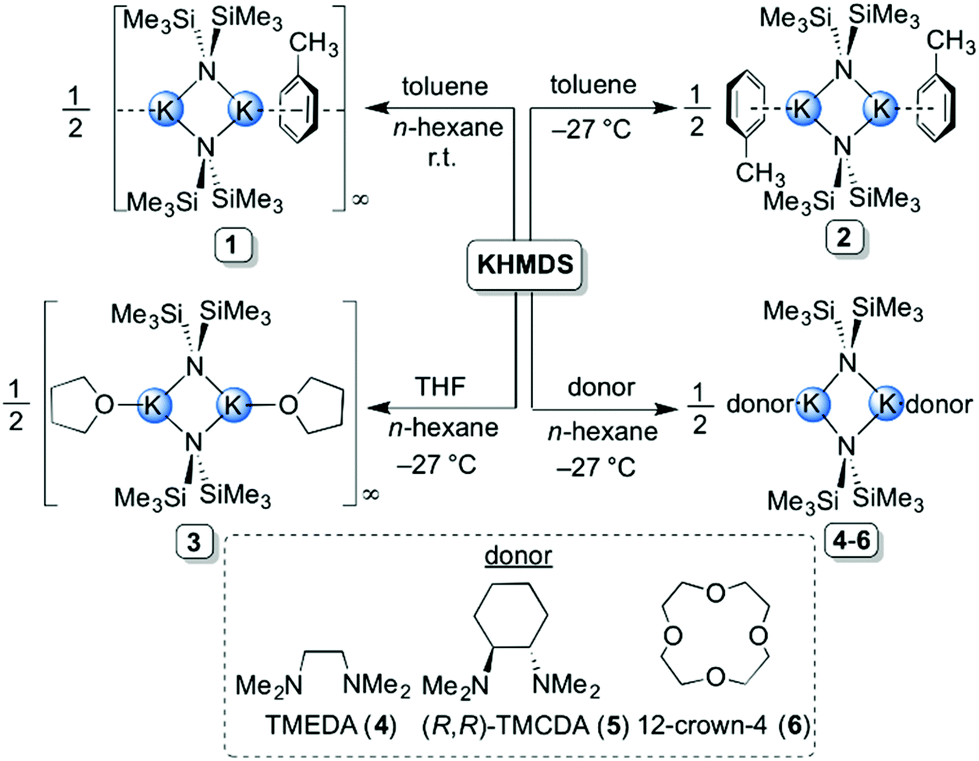
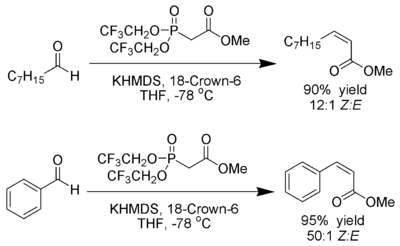
Exploring the solid state and solution structural chemistry of the utility amide potassium hexamethyldisilazide (KHMDS) - Dalton Transactions (RSC Publishing) DOI:10.1039/C7DT01118K
![Asymmetric total synthesis of (−)-lingzhiol via a Rh-catalysed [3+2] cycloaddition | Nature Communications Asymmetric total synthesis of (−)-lingzhiol via a Rh-catalysed [3+2] cycloaddition | Nature Communications](https://media.springernature.com/lw685/springer-static/image/art%3A10.1038%2Fncomms6707/MediaObjects/41467_2014_Article_BFncomms6707_Fig1_HTML.jpg)
Asymmetric total synthesis of (−)-lingzhiol via a Rh-catalysed [3+2] cycloaddition | Nature Communications
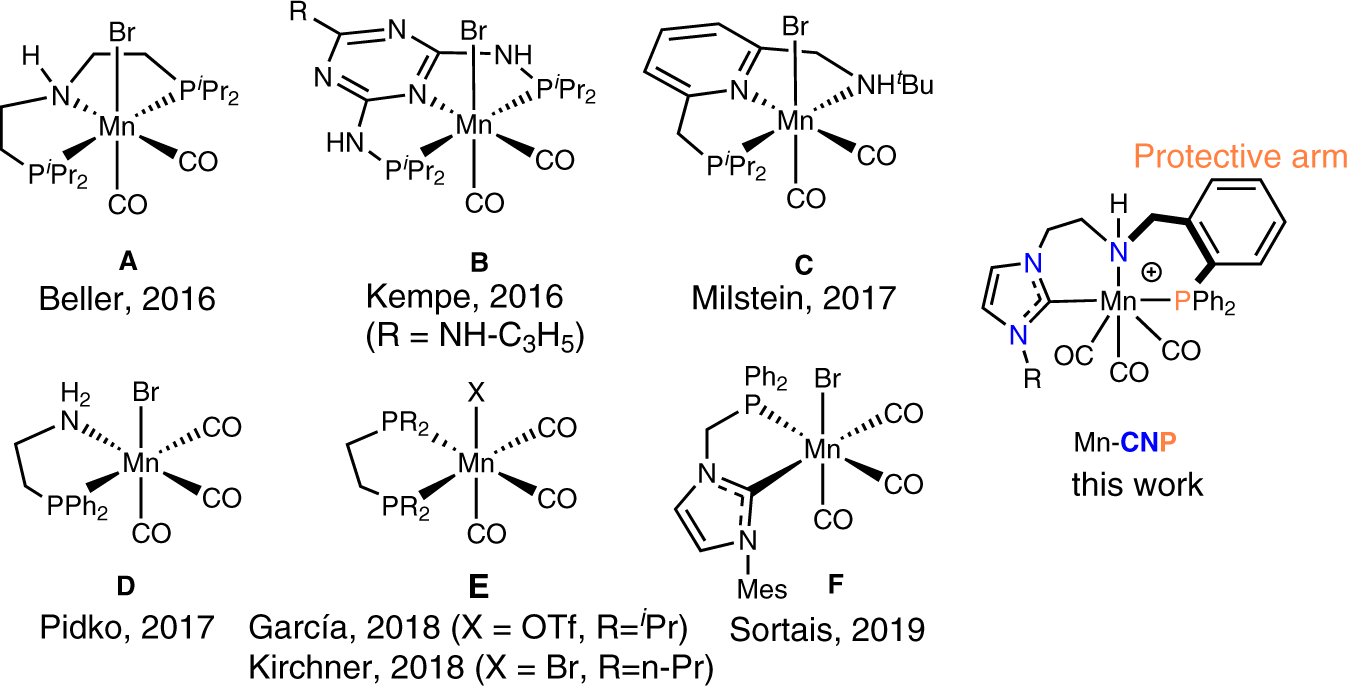
Robust and efficient hydrogenation of carbonyl compounds catalysed by mixed donor Mn(I) pincer complexes | Nature Communications
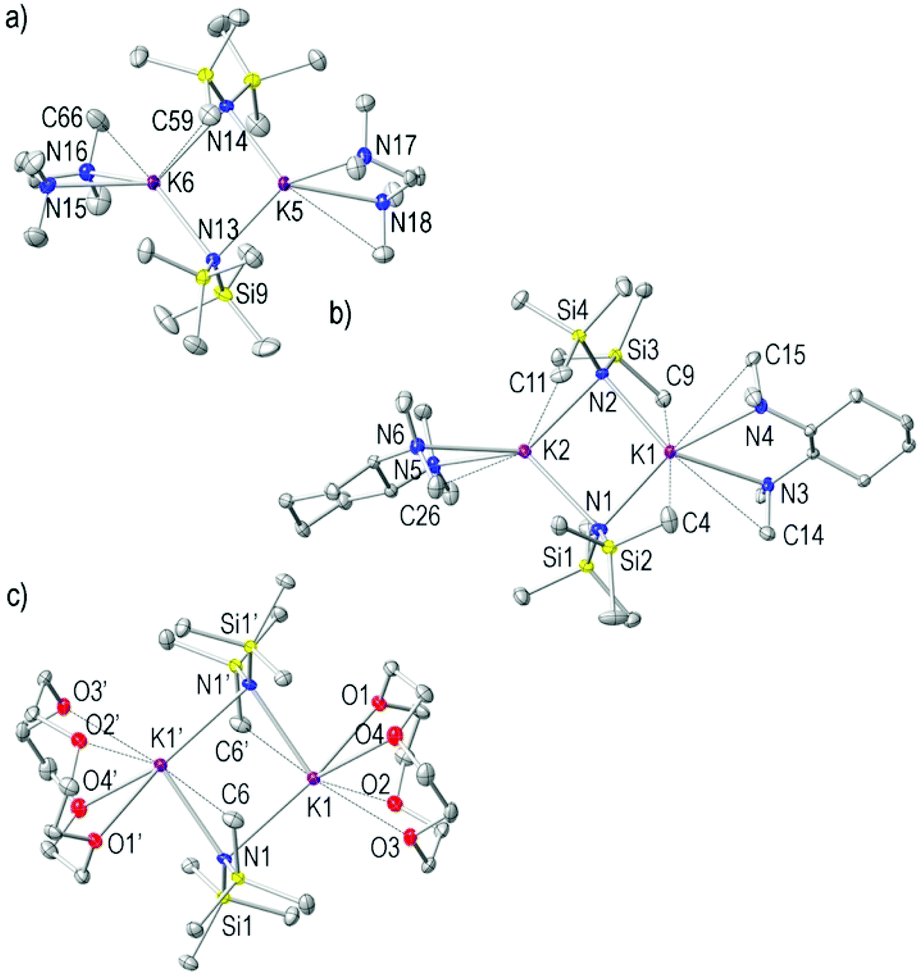
Exploring the solid state and solution structural chemistry of the utility amide potassium hexamethyldisilazide (KHMDS) - Dalton Transactions (RSC Publishing) DOI:10.1039/C7DT01118K
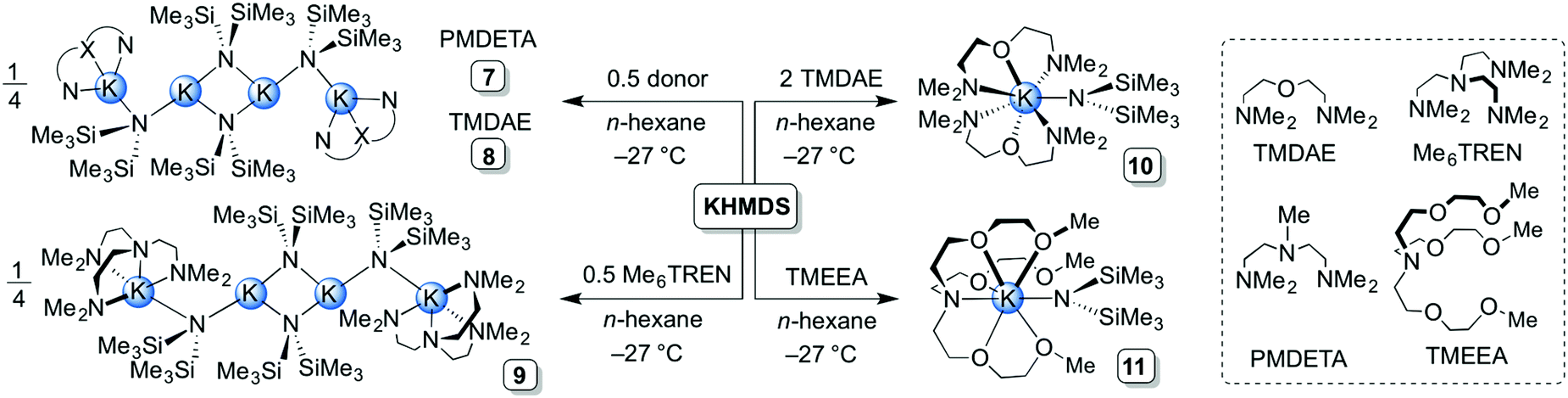
Exploring the solid state and solution structural chemistry of the utility amide potassium hexamethyldisilazide (KHMDS) - Dalton Transactions (RSC Publishing) DOI:10.1039/C7DT01118K